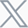A Conceptual Shift in Metabolic Health Optimization
In weight management, reduction in body weight has long been regarded as the most intuitive and important endpoint. However, with the advancement of obesity research and clinical practice, it has become increasingly clear that body weight alone does not fully capture the underlying metabolic state of an organism. Particularly in individuals with obesity or metabolic dysfunction, weight loss may arise from fundamentally different physiological processes, leading to divergent health outcomes.
Many weight-loss interventions—whether pharmacological or lifestyle-based—result not only in reductions in fat mass but also in losses of lean body mass, especially skeletal muscle. Skeletal muscle plays a central role in maintaining physical function and represents a major site for glucose uptake and energy expenditure. Declines in muscle mass are associated with reductions in basal metabolic rate and may exacerbate insulin resistance, contributing to weight regain, fatigue, and worsening metabolic status in certain individuals.
Therefore, weight reduction is not the ultimate goal; rather, the composition of weight loss is of primary importance. When weight reduction predominantly reflects fat mass loss, metabolic risk is generally improved. In contrast, when weight loss occurs at the expense of lean mass, its long-term health value must be carefully reconsidered.
Considering this, fat reduction with preservation or enhancement of lean mass has emerged as a key objective in next-generation anti-obesity drug development.
Evaluation Upgrade: A Multidimensional Framework for Next-Generation Therapeutics
From a drug development perspective, the concept of fat loss with muscle preservation represents a measurable and data-driven evaluation framework. If preclinical studies rely solely on body weight as a primary endpoint, it becomes difficult to distinguish whether weight reduction results from improved metabolic efficiency or from excessive appetite suppression, intolerance, or potential toxicity. The former reflects genuine therapeutic potential, whereas the latter may pose safety and compliance concerns in clinical settings.
Consequently, an increasing number of anti-obesity programs incorporate multidimensional endpoints during the preclinical phase, including body composition analysis, metabolic profiling, and behavioral assessments. Integrated evaluation across these parameters enables early identification of compound effects on adipose tissue, skeletal muscle, and systemic metabolism, thereby supporting dose selection, combination strategies, and clinical positioning.
GemPharmatech Empowerment: Enabling Innovative Research Through Advanced Disease Models
As an innovator in laboratory animal models, GemPharmatech provides high-quality genetically engineered rodent models and comprehensive technical services to academic institutions and pharmaceutical companies worldwide. Our portfolio includes diet-induced obesity (DIO) models and related metabolic disease models in both mice and rats, enabling robust simulation of human metabolic disorders. Through advanced gene-editing technologies, we provide drug target humanized models to facilitate mechanistic exploration of anti-obesity therapeutics. In addition, we offer integrated services including body composition analysis and energy metabolism assessment, enabling precise evaluation of drug effects across adipose tissue, skeletal muscle, and multiple metabolic organs. From model selection to data interpretation, we provide end-to-end solutions to accelerate translational research.
Our Integrated Model Platform Accelerating Translational Innovation
| Strain Type | Strain Name | ||
| Diet-induced obesity (DIO) model | • B6 DIO RD • SD DIO 60% | ||
| Genetically engineered spontaneous obesity model | • B6-ob • B6-Chr1 YP1 • B6-Alms1-del • BKS-db | ||
| Humanized strains | • B6-hGLP-1R • B6-hGIPR • B6-hGCGR • B6-hGIPR/GCGR • B6-hGLP-1R/GIPR • B6-hGLP-1R/GCGR • B6-hGLP-1R/GIPR/GCGR • B6-Glp1r(S33W) • SD-Glp1r(S33W) | • B6-hACVR1C (ALK7) • B6-hINHBE • B6-hMSTN • B6-hGPR75 • B6-hMTARC1 • BKS-hGRB14 • B6-hDGAT2 • B6-hMC4R • B6-hTFRC/GPR75 | • B6-hALK4 • B6-hALK5 • B6-hACVR2A • B6-hACVR2B • B6-hTFRC/ACVR2A • B6-hTFRC/ACVR2B • B6-ACVR2A/ACVR2B • B6-hTFRC/ACVR2A/ACVR2B |
Representative Case Studies
Case 1: Semaglutide and Bimagrumab in a DIO Mouse Model
Combination therapy with Bimagrumab and Semaglutide reduced adipose tissue mass while preventing lean tissue loss in DIO mice.
Semaglutide reduced body weight through decreases in both fat and lean mass.
Bimagrumab reduced fat mass while increasing lean mass in DIO mice.
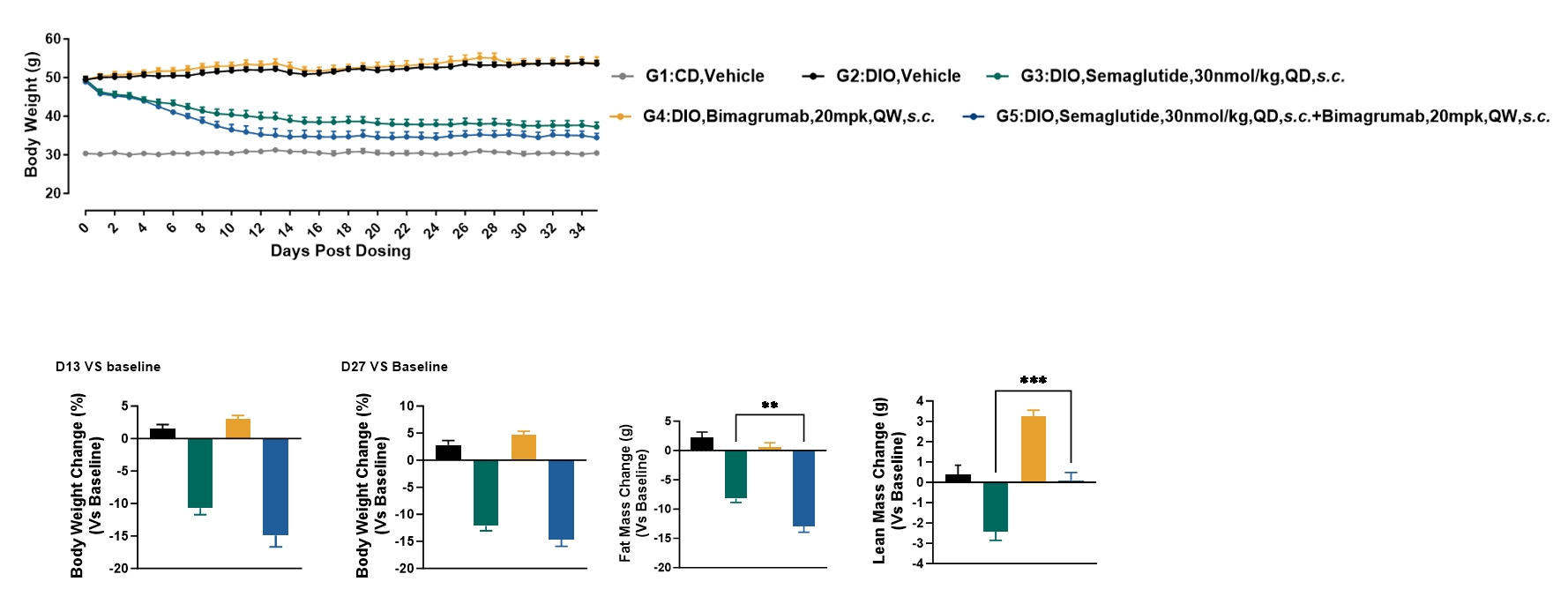
Case 2: Cagrilintide and Eloralintide in a DIO Rat Model
Both Cagrilintide and Eloralintide significantly reduced body weight and food intake in DIO rats.
Weight loss induced by Cagrilintide was driven by reductions in both fat and lean mass.
Weight loss induced by Eloralintide was primarily attributable to fat mass reduction.
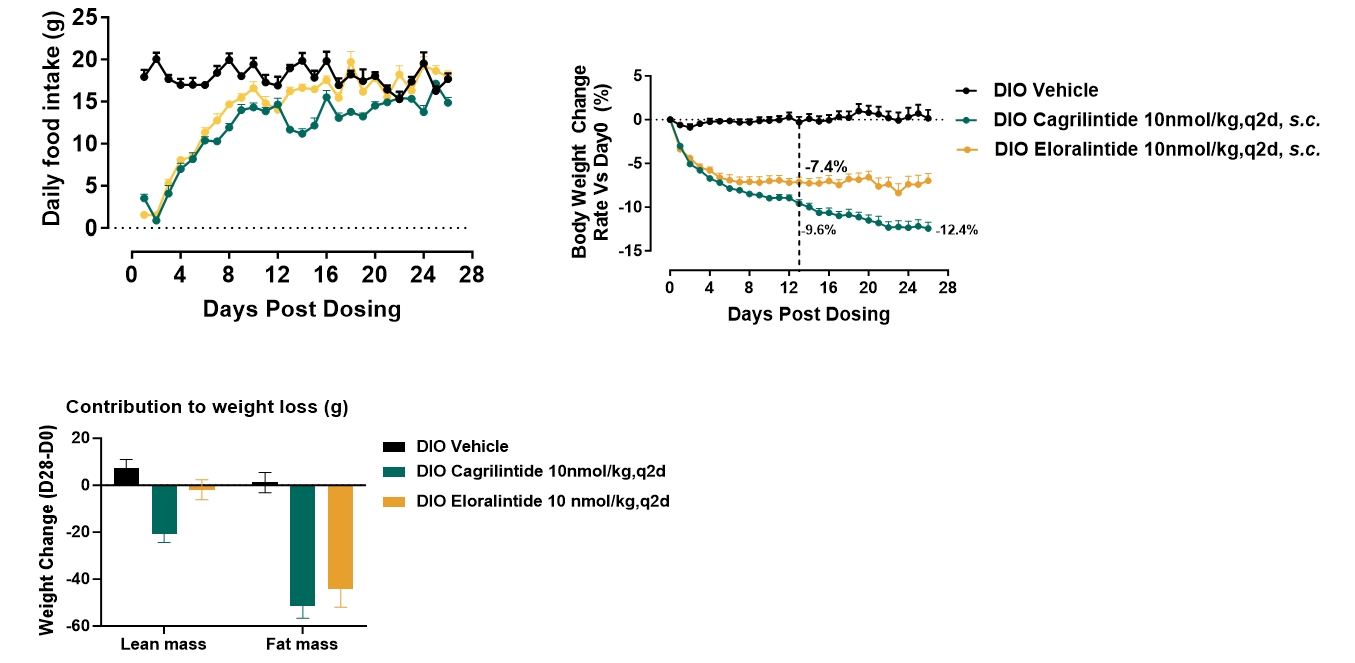
Case 3: Orforglipron in a GLP-1R Humanized DIO Mouse Model
Orforglipron significantly reduced food intake in GLP-1R humanized mice.
In high-fat diet–induced hGLP-1R mice, both Orforglipron and Semaglutide significantly reduced body weight and food intake, and improved glucose tolerance.
Validated Benchmark Therapeutics within the DIO Platform
Therapeutic Modality | Validated Compounds | Mechanism of Action |
Peptides | Semaglutide | GLP-1R agonist |
Tirzepatide | GLP-1R/GIPR dual agonist | |
Mazdutide | GLP-1R/GCGR dual agonist | |
Retatrutide | GLP-1R/GIPR/GCGR triple agonist | |
Cagrilintide | AMYRs/CTR agonist | |
Eloralintide | AMYRs agonist | |
Rybelsus | Oral GLP-1R agonist | |
Antibodies | Anti-GIPR | GIPR antagonist |
Bimagrumab | ACVR2A/B antagonist | |
SiRNA | siRNA-INHBE | INHBE gene silencing |
siRNA-ALK7 | ALK7 gene silencing | |
Small Molecules | MGL-3196 | THR-β agonist |
Orforglipron | GLP-1R agonist |
In summary, the paradigm of anti-obesity drug development is rapidly evolving from a sole focus on body weight reduction toward a more sophisticated and clinically meaningful objective—achieving fat mass reduction while preserving or enhancing lean mass. This shift reflects a deeper understanding of metabolic health, long-term treatment sustainability, and patient outcomes. By integrating multidimensional evaluation endpoints and leveraging advanced preclinical model platforms, researchers can more accurately characterize therapeutic potential and de-risk clinical translation. Through comprehensive disease models and translationally relevant evaluation systems, GemPharmatech is committed to supporting next-generation metabolic innovation and accelerating the development of safer, more effective therapies.
References
1. Levin BE, Geary N, Lutz TA. The (dys)regulation of energy storage in obesity. Physiol Rev. 2025;105(3):803–895.
2. Muzurović E, et al. Emerging incretin- and multi-agonist-based treatments for cardio-kidney-liver-metabolic diseases. Metabolism. 2026.