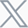Accelerating T-Cell Engager (TCE) Development: Advanced Preclinical Evaluation Solutions

In recent years, the landscape of cancer immunotherapy has undergone a paradigm shift across diverse oncological indications. However, achieving robust efficacy in solid tumors remains a significant challenge, often due to downregulated MHC molecules or insufficient immune cell infiltration within the tumor microenvironment (TME). While autologous CAR-T cell therapies offer potent cytotoxicity, their widespread adoption is frequently hampered by lengthy manufacturing timelines, high production costs, and the complexities of personalized medicine.
The Strategic Shift Toward T-Cell Engagers (TCEs)
Compared to cell-based therapies, TCEs—standardized, “off-the-shelf” antibody-based therapeutics—offer distinct operational advantages, including shorter development cycles, scalable manufacturing, and superior batch-to-batch consistency. These attributes make TCEs highly favorable for large-scale clinical applications, positioning them as a critical immunotherapeutic pillar alongside immune checkpoint inhibitors and CAR-T therapies.
By physically bridging T cells and tumor cells, TCEs facilitate the recruitment and activation of endogenous T cells in an MHC independent manner. This provides a key mechanistic advantage for overcoming the immunosuppressive TME (Figure 1).
The Evolution into Solid Tumor Indications
To date, 11 TCE therapeutics have received global regulatory approval. The recent approval of Tarlatamab-dlle as the first TCE for small cell lung cancer (SCLC) represents a landmark achievement, signaling the successful expansion of TCE technology from hematological malignancies to solid tumors.
Current clinical development in solid tumors is accelerating, with SCLC and metastatic castration-resistant prostate cancer (mCRPC) leading the way. Key targets such as DLL3, STEAP1, and PSMA are at the forefront, with multiple Phase III trials currently in progress.
Overcoming Translational Bottlenecks
Despite their potential, conventional TCEs face several translational hurdles, including:
•Safety risks: Cytokine Release Syndrome (CRS) and off-target toxicity.
•Pharmacokinetics: Short serum half-life.
•Efficacy barriers: Poor tumor penetration and antigen heterogeneity.
To address these challenges, current research focuses on identifying highly specific targets and exploring intracellular antigens via TCR-mimic strategies. Modulating CD3 affinity and utilizing “masked” or conditionally activated designs are also promising approaches to widen therapeutic window. Furthermore, strategies such as half-life extension, local mRNA delivery, and combination regimens with chemotherapy or other immunotherapies are being employed to optimize clinical outcomes.
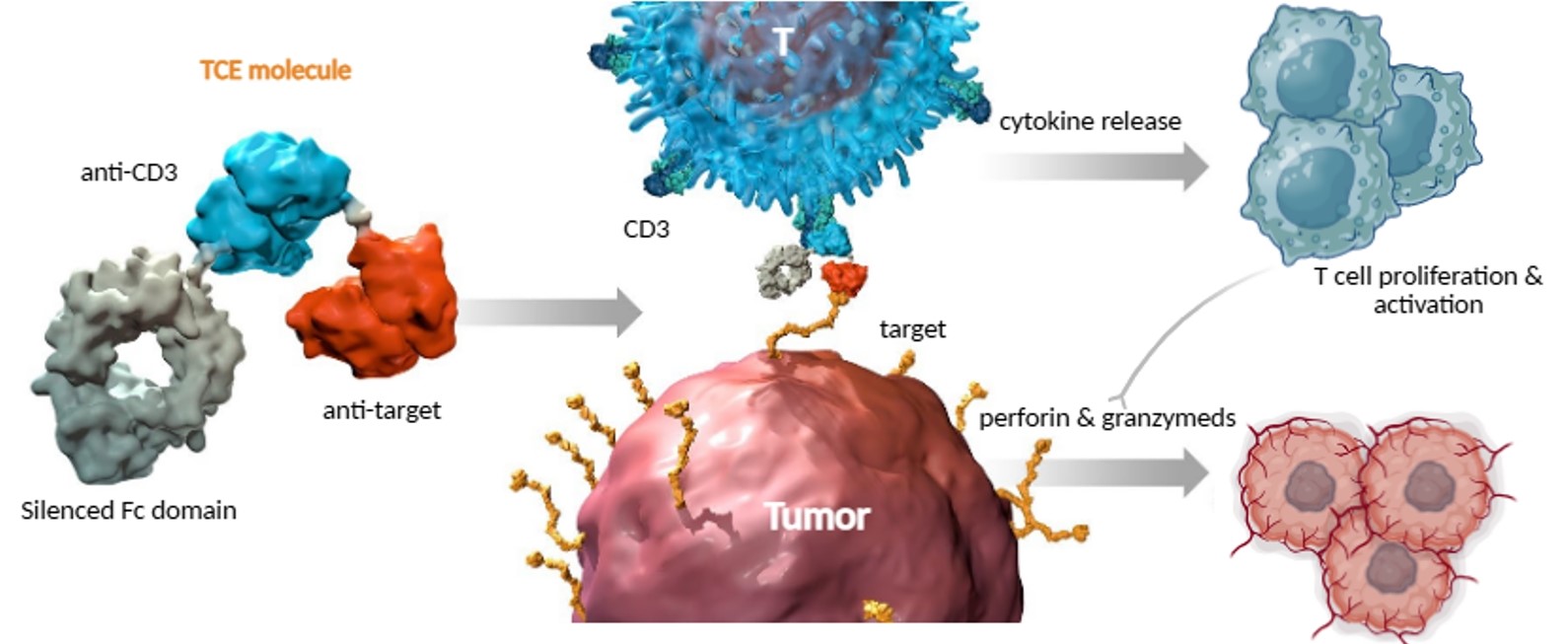
Figure 1. Mechanism of TCE Therapeutics
GemPharmatech’s Integrated Preclinical Evaluation Platform
Success in TCE development hinges on rigorous non-clinical efficacy and safety validation. GemPharmatech offers a comprehensive, end-to-end preclinical evaluation solution designed to navigate the complexities of model selection, humanization, and donor variability. Contact Us
In Vitro Pharmacodynamics
Our in vitro platform provides high-quality data across the entire early-stage development workflow:
Antigen characterization: Precise antigen quantification and binding affinity assays.
T-cell functionality: Comprehensive assessment of T-cell activation and proliferation.
Cytotoxicity assays: Standard TDCC (T-cell dependent cellular cytotoxicity), re-challenge assays, and bystander killing evaluations.
Safety screening: Cytokine release profiling using bead-based multiplex assays (e.g., CBA).
Advanced In Vivo Efficacy Models
GemPharmatech leverages a diverse suite of proprietary mouse models to support varied MOA requirements:
System | Model Basis | Key Advantages | Primary Application |
huPBMC-NCG | Human PBMC | Rapid reconstitution (7–10 days); 200+ donor library | Rapid efficacy screening |
huPBMC-NCG-MHC-dKO | MHC-I/II Double Knockout | Significantly delays GvHD onset (>10 weeks), extending the therapeutic window | Long-term efficacy assessment |
huHSC-NCG | CD34+ huHSC | Reconstitutes a human immune system (T, B, NK, and myeloid cells) | Comprehensive efficacy and non-GLP safety |
hCD3 Mice | Humanized CD3ϵ/δ/γ | Dual-background models with 190+ humanized TAA (Tumor Associated Antigen) targets | Mechanism of action and safety/toxicity studies |
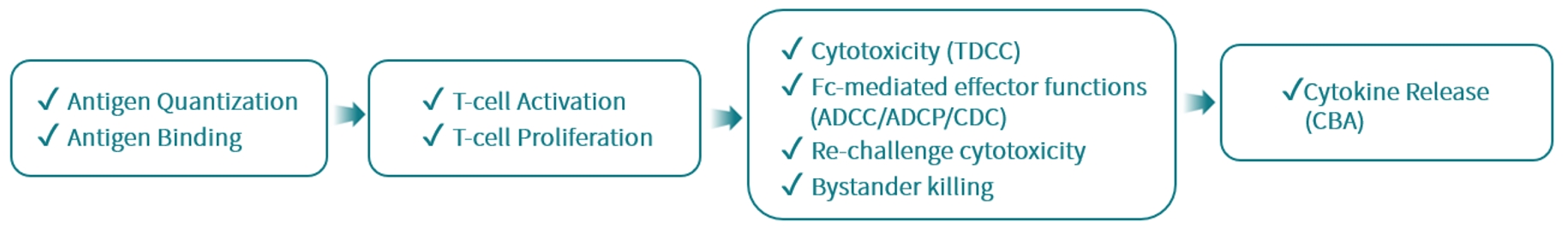
Case Study: Pharmacodynamic Evaluation of Xaluritamig (CD3/STEAP1)
In a validation study of Xaluritamig, our platform demonstrated high sensitivity and translational relevance:
In Vitro: The agent elicited potent, dose-dependent TDCC activity and cytokine release in STEAP1-expressing cell lines.
In Vivo: Significant tumor growth inhibition (TGI) was observed in both STEAP1 high-expression (LNCaP clone FGC) and low-expression (22RV1) tumor models.
Safety: Profiling confirmed that immune activation was localized to the tumor microenvironment, indicating a low risk of systemic CRS at the tested doses.
Figure 2. In Vitro and In Vivo Pharmacodynamic Evaluation of Xaluritamig
A: Xaluritamig elicited potent TDCC activity in vitro. B: Xaluritamig demonstrated significant tumor growth inhibition in LNCaP clone FGC (STEAP1 high-expression) tumors. C: Xaluritamig showed significant tumor growth inhibition in 22RV1 (STEAP1 low-expression) tumors.
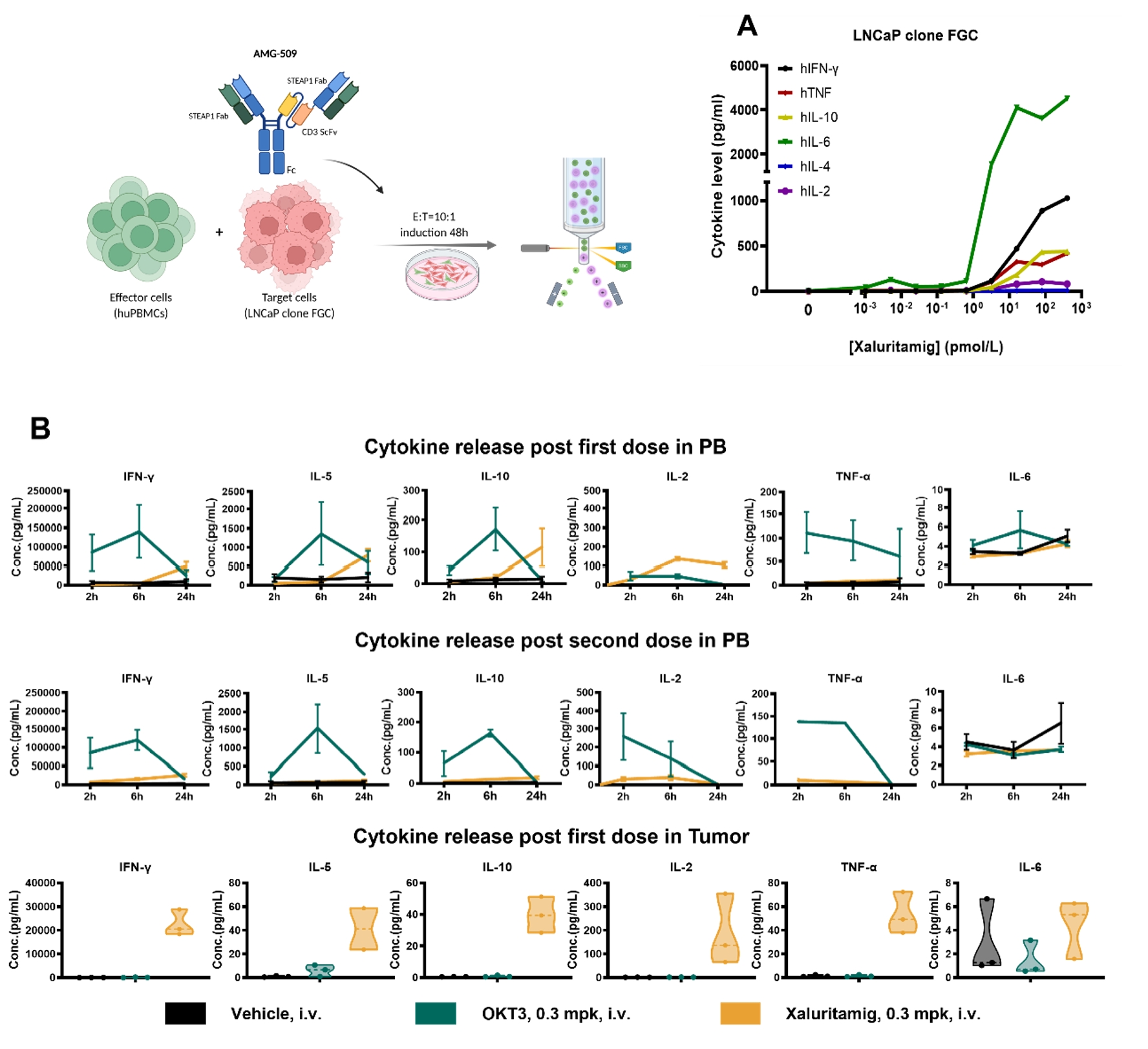
Figure 3. In Vitro and In Vivo Cytokine Release Induced by Xaluritamig
A: Xaluritamig elicited cytokine release in vitro. B: Xaluritamig induced minimal systemic cytokine release but high local cytokine levels within the tumor microenvironment.
Why Partner with GemPharmatech?
GemPharmatech provides the infrastructure and expertise necessary to accelerate your TCE program from discovery to IND filing.
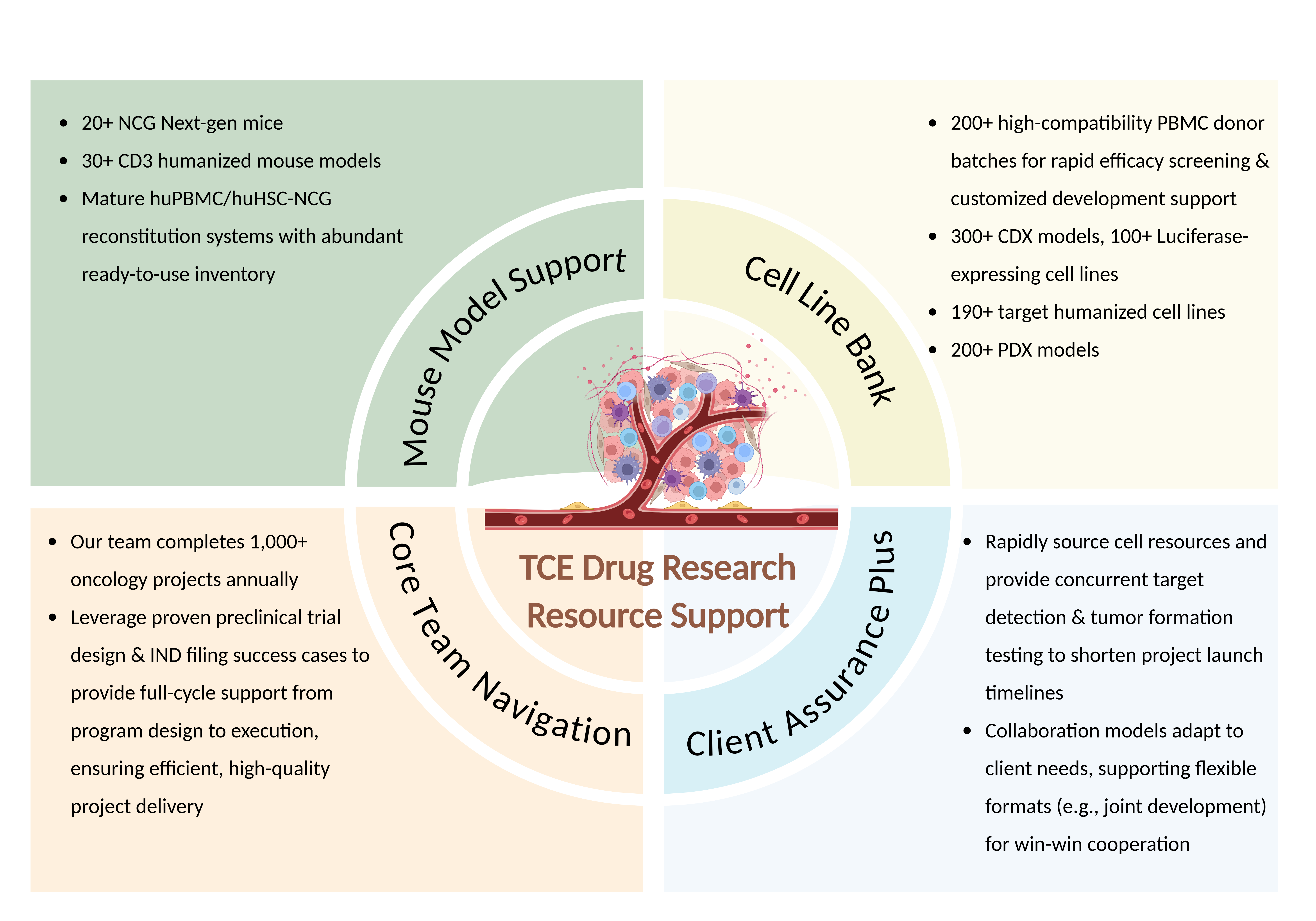
Don't Let Translational Hurdles Slow You Down.
GemPharmatech's extensive library of humanized models and TAA targets can help you de-risk your TCE program and accelerate its path to the clinic.
Contact our experts today to discover how we can support your next milestone.