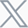Overcoming Obesity: Challenges Facing Weight Loss Drugs and the Promise of Combination Therapies

In the ongoing battle against obesity, the development of pharmaceutical drugs such as Semaglutide, a glucagon-like peptide-1 receptor agonist (GLP-1RA), has shown promising results in enhancing weight loss and improving metabolic health. However, its use at high doses often leads to poor tolerance and gastrointestinal side effects among patients, which can result in treatment discontinuation. In addition, there is growing concern about the association between Semaglutide use and muscle loss, presenting another hurdle in its therapeutic application for effective obesity management.
To tackle these issues, several innovative solutions are being pursued by researchers. Currently, one such focus lies in the use of combination therapies. The objective of such strategies is to improve a patient’s weight loss outcome and/or reduce the adverse effects associated with GLP-1RAs. Several encouraging developments in new pharmacological combinations targeting various pathways involved in weight regulation have recently emerged. For instance, the combination of GLP-1RAs with other entero-pancreatic hormones such as gastric inhibitory peptide (GIP), glucagon, or amylin, enhances the effects of GLP-1RA on weight loss and metabolism1. Another example includes the use of medications that modulate an individual’s total daily energy expenditure (TDEE). As the body’s basal metabolic rate (BMR) contributes to about 60% to 75% of an individual’s TDEE, medications such as THR-β agonists that modulate TDEE could amplify the weight loss effects of GLP-1RAs. Indeed, preclinical studies have indicated combining THR-β agonists with GLP-1RA improves body weight loss and while preserving lean mass in diet-induced obesity (DIO) mice2. Lastly, using rodent obesity models, activin type II receptor blockers like Bimagrumab have demonstrated efficacy in mitigating muscle loss associated with Semaglutide treatment3. Currently, clinical trials are ongoing to explore the safety and efficacy of the Semaglutide and Bimagrumab combination in adult obesity patients4.
Rodent obesity models play an important role in the drug evaluation process and offer exciting new avenues for research in the weight loss space. Such models enable essential insights into the pharmacodynamics and safety profiles of novel combinations before advancing to human trials. GemPharmatech has established a validated obesity efficacy testing system built upon a high-quality, large-scale 'ready-to-use' DIO mice colony and a comprehensive and validated in vivo and in vitro testing platform.
Efficacy Study of Semaglutide and Bimagrumab in DIO Mice
To evaluate whether a combination of Bimagrumab and Semaglutide can preserve/increase lean muscle mass while retaining weight and/or fat mass loss, DIO mice (>50g) were treated with Semaglutide, Bimagrumab or a combination of both for two weeks via subcutaneous injections given either daily or weekly. Body weight, food intake, white adipose tissue (WAT) and muscle mass were measured throughout the study. The results demonstrated that Bimagrumab not only amplified Semaglutide induced body weight loss (Figure1), but also attenuated lean mass loss associated with Semaglutide’s (Figure2).
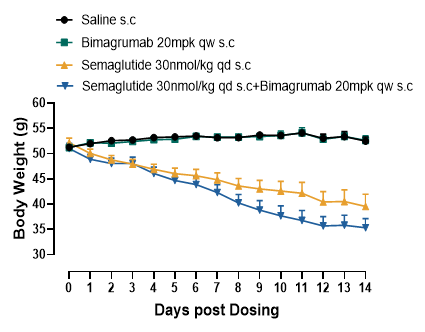
Figure 1. Combination treatment of Bimagrumab and Semaglutide showed greater weight loss than Semaglutide single treatment group. Data were presented as Mean±SEM.
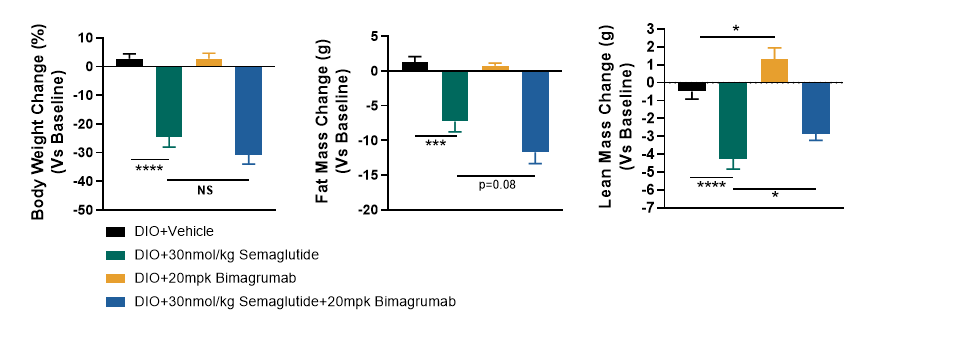
Figure 2. Combination treatment of Bimagrumab and Semaglutide partially rescued Semaglutide’s association with lean mass loss and amplified fat mass loss. Data were presented as Mean±SEM. ns, not significant;. *, p<0.05; ***, p<0.001; ****, p<0.0001 by one way ANONA.
Efficacy Study of Semaglutide and MGL-3196 in DIO Mice
To determine whether the themogenesis effect of MGL-3196 could increased the body weight loss effect of Semaglutide, DIO mice (>50g) were treated with Semaglutide, MGL-3196 or a combination of both for 4 weeks. The body weight, food intake, body composition, glucose tolerance and energy expenditure were measured. The results showed that MGL-3196 significantly enhanced semaglutide induced body weight loss without affecting food intake (Figure3 and Figure4). Compared with the single treatment group of Semaglutide, the combination treatment group of Semaglutide and MGL-3196 further reduced fat mass while preserving lean mass (Figure5). Additionally, glucose intolerance was notably improved in the combination treatment compared to the Semaglutide single treatment group (Figure6). Moreover, the results of calorimetry analysis using Comprehensive Lab Animal Monitoring System (CLAMS) suggested that the combination of MGL-3196 and Semaglutide was able to enhance energy expenditure of DIO mice (Figure7).
Figure 3. Semgalutide and MGL-3196 combination treatment enhanced body weight loss in DIO mice. Data were presented as Mean±SEM.
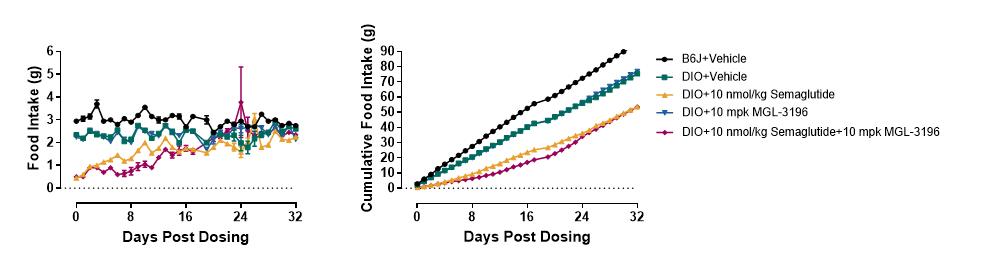
Figure 4. MGL-3196 did not affect food intake in DIO mice. Data were presented as Mean±SEM.
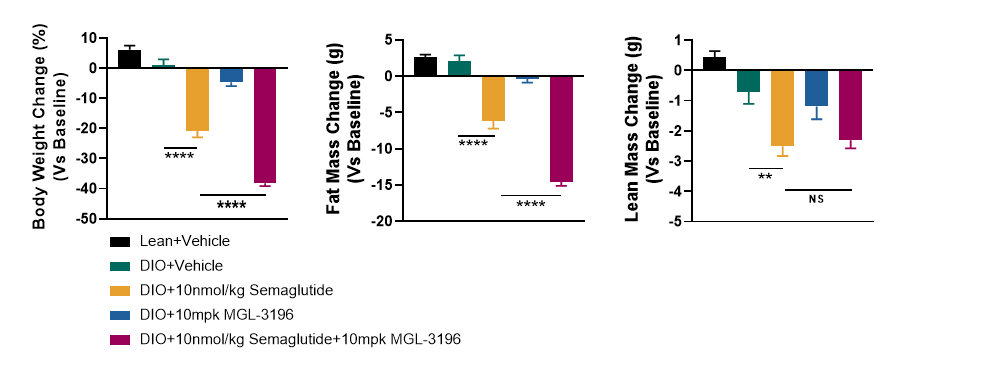
Figure 5. Combination treatment of MGL-3196 and Semaglutide significantly amplified the decline of fat mass in DIO mice while preserving lean mass. Data were presented as Mean±SEM. ns, not significant; **, p<0.01; ****, p<0.0001 by one way ANONA.
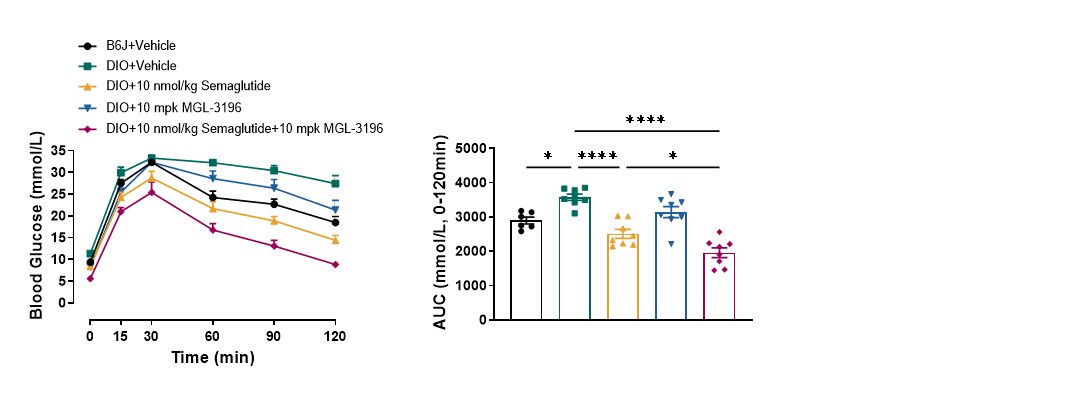
Figure 6. Combination treatment of MGL-3196 and Semaglutide significantly amplified the improvement of glucose tolerance in DIO mice. Data were presented as Mean±SEM. *, p<0.05; ****, p<0.0001 by one way ANONA.
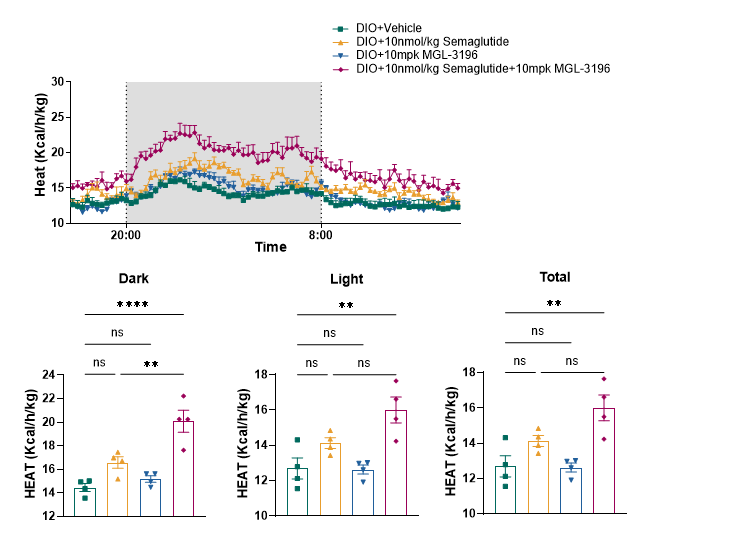
Figure 7. MGL-3196 enhanced energy expenditure in DIO mice. Data were presented as Mean±SEM. ns, not significant; **, p<0.01; ****, p<0.0001 by one way ANONA.
Reference
1. Melson E, Ashraf U, Papamargaritis D, Davies MJ. What is the pipeline for future medications for obesity? Int J Obes (Lond). 2024 Feb 1. doi: 10.1038/s41366-024-01473-y. Epub ahead of print. PMID: 38302593.
2. https://ir.ternspharma.com/news-releases/news-release-details/terns-pharmaceuticals-highlights-new-preclinical-data-supporting/
3. Nunn E, Jaiswal N, Gavin M, Uehara K, Stefkovich M, Drareni K, Calhoun R, Lee M, Holman CD, Baur JA, Seale P, Titchenell PM. Antibody blockade of activin type II receptors preserves skeletal muscle mass and enhances fat loss during GLP-1 receptor agonism. Mol Metab. 2024 Feb;80:101880. doi: 10.1016/j.molmet.2024.101880. Epub 2024 Jan 11. PMID: 38218536; PMCID: PMC10832506.
4. https://clinicaltrials.gov/study/NCT05616013